Fully compliant UX and software development.
We provide UX and development services that comply 100% with IEC 62304 and IEC 62366.

Software Development
Fully IEC 62304 compliant development.
We provide fully compliant documentation.
Integration with your eQMS.
QA testing (verification) and documentation.
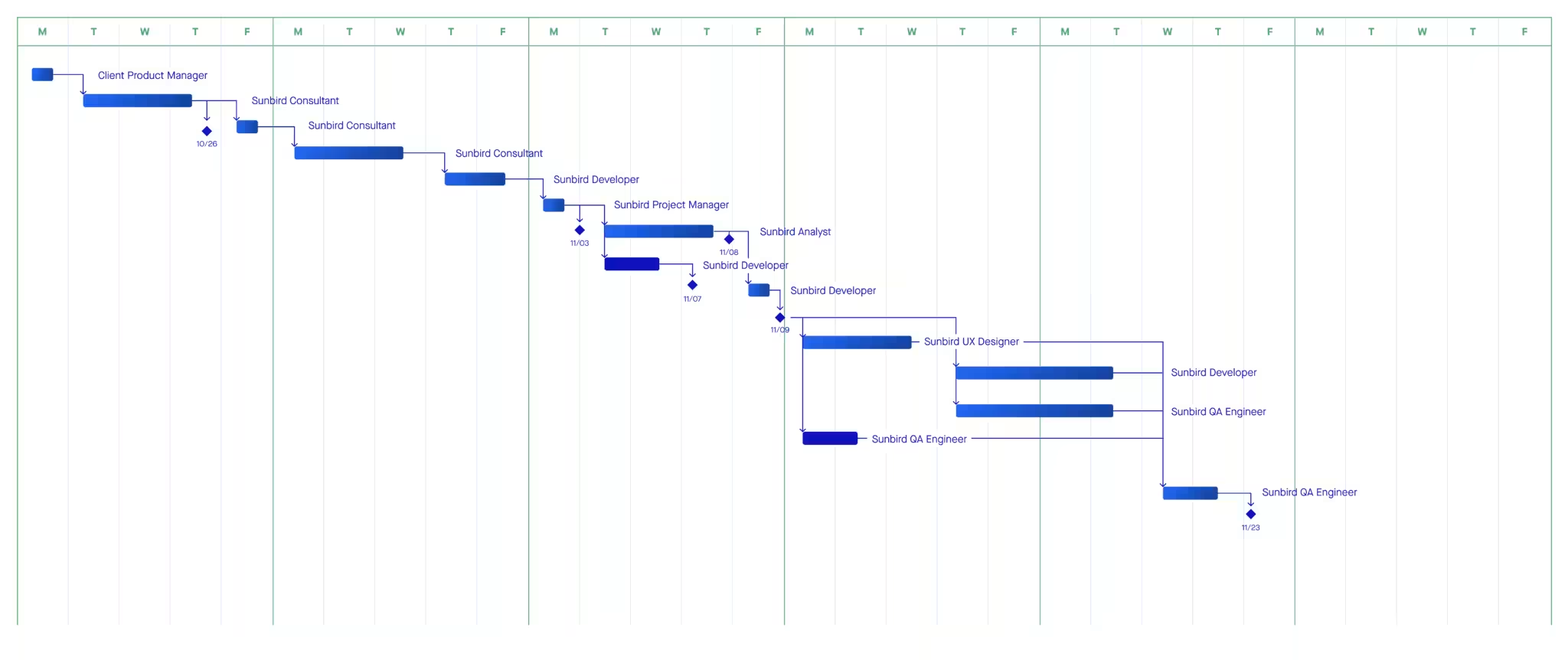
PORTFOLIO
We helped to set up their first development processes and documentation, allowing them a quick start in compliant software development.
We helped to increase of development velocity by simplifying their SOP’s, reducing documentation burdens and improving processes.
We helped Replasia start their journey towards a certified SaMD medical device.
We provide fully compliant documentation
For all our clients, we either create compliant documentation or set up the client for producing compliant documentation. We also provide trainings on this subject.
Software documents:
- Software Development Plan
- Software Requirements Specification
- Software Architecture Document
Verification & risk documents:
- Software Verification Plan
- Software Verification Report
- Software Risk Management Plan
SOP documents:
- SOP Design & Development
- SOP Software Release
- SOP Software Problem Resolution
We seamlessly connect with your QMS
Implementing a Quality Management System (QMS) in the development flow of an organization can help ensure that the final product meets the customer’s needs and expectations. Our development flow seamlessly integrates with a QMS, making it easier to manage quality and reduce risk throughout the development process.
- Our development process utilizes rigorous testing and verification procedures to ensure quality
- Regular quality audits are conducted to identify areas for improvement and verify compliance with regulations and standards
- Continuous improvement principles are integrated into our development flow to refine processes and improve quality over time
We provide QA testing (verification) and documentation
Our QA testing and documentation services ensure that your product meets the highest quality standards and is fully documented to meet regulatory requirements.
Ready to go next level?
- Get up to speed quickly with required documentation
- Accelerate your market submission
- Increase efficiency of your software engineering team

UX Consulting
We offer comprehensive support in these areas to ensure that our clients’ products are intuitive, user-friendly, and meet the needs of healthcare professionals and patients. Our team of software engineers and UX designers work closely with clients to develop solutions that enhance the usability, reliability, and effectiveness of their products, in regulatory compliance

What we offer
- Conducting user research to understand the needs and goals of users and identifying areas for improvement in the user experience of the medical device software.
- Developing wireframes, mockups, and prototypes to design an intuitive and effective user interface that meets the needs of users and provides a positive user experience.
- Developing wireframes, mockups, and prototypes to design an intuitive and effective user interface that meets the needs of users and provides a positive user experience.
Why?
- To validate ideas and concepts
- To evaluate the current product
- To make informed decisions for developing new features
- To have a plan before starting the development process
- To develop a product that is understood by your target audience
- To reduce ineffective development cycles
- To build a future-proof product with a design system
- To make sure your software meets regulatory requirements
- To back up your risk assessments with real data